Pediatric congestive cardiomyopathy
Author arrhythmia
2019-04-21
Congestive cardiomyopathy, also called dilated cardiomyopathy (DKM), is characterized by stretching and weakening of the heart muscle, which cannot effectively pump blood. It is often combined with heart failure, which occurs when the heart is not sufficiently reduced to meet the needs of the body. If the heart does not work with the necessary force, the tissues and organs do not receive enough oxygen.
About 5.8 million people in the United States suffer from heart failure, and about 700,000 new cases are diagnosed each year.
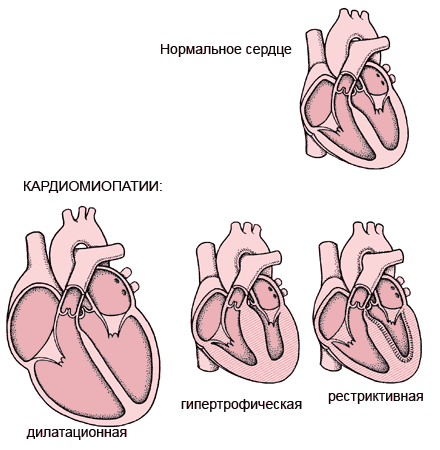
Cardiomyopathy can be considered a cause of heart failure. There are two main types of cardiomyopathy: primary cardiomyopathy, defined as changes in the structure or function of the heart muscle, which cannot be attributed to a specific reason, and secondary, associated with impaired functioning of the heart or other organs. Congestive cardiomyopathy is the most common primary form of heart muscle disease. Other types include hypertrophic and restrictive cardiomyopathy.
Video: Dilated (Congestive) Cardiomyopathy
Causes of congestive cardiomyopathy in children
Myocardial damage, followed by the development of cardiomyopathy, can occur for various reasons. However, in the overwhelming majority of patients, the specific etiology is not defined (i.e., idiopathic DCM occurs).
Three main factors were involved in the pathogenesis of myocardial damage in DCM:
- Previous viral myocarditis.
- Autoimmune disease.
- Genetic predisposition.
Factors identified as causes of myocardial damage and the development of congestive cardiomyopathy in children
| Factor category | Detailed development factors |
| Viral infections | Coxsackie B virus, human immunodeficiency virus, echovirus, rubella, chickenpox, parotitis, Epstein-Barr virus, cytomegalovirus, measles, polio. |
| Bacterial infections | Diphtheria, mycoplasma, tuberculosis, Lyme disease, septicemia. |
| Rickettsia | Psittacosis, spotty fever of rocky mountains. |
| Parasites | Toxoplasma, toxocar, cysticerc. |
| Mushrooms | Histoplasma, coccidioidomycosis, actinomycetes. |
| Neuromuscular disorders | Duchenne or Becker muscular dystrophies, Friedreich's ataxia, Kearns-Sayre syndrome, other muscular dystrophies. |
| Nutritional factors | Kwashiorkor, pellagra, thiamine deficiency, selenium deficiency. |
| Collagen Vascular Diseases | Rheumatism, rheumatoid arthritis, systemic lupus erythematosus, dermatomyositis, Kawasaki disease. |
| Hematological diseases | Thalassemia, sickle cell anemia, iron deficiency anemia. |
| Coronary heart disease | Abnormal left coronary artery, myocardial infarction. |
| Medicines | Anthracycline, cyclophosphamide, chloroquine, iron overload. |
| Endocrine diseases | Hypothyroidism, hyperthyroidism, hypoparathyroidism, pheochromocytoma, hypoglycemia. |
| Metabolic disorders | Diseases of glycogen accumulation, carnitine deficiency, fatty acid oxidation defects, mucopolysaccharidoses. |
| Malformations | Cry-Doo-Chat Syndrome (feline cry) |
Viral myocarditis
Diverse studies revealed signs of enterovirus infection, in particular the Coxsackie virus, in 20-25% of patients. Also various other viruses are often identified. In fact, the most common associated viruses change over time.
Currently, the Coxsackie B virus is probably the less common cause of the development of DKM than in the past. So far, no methods can be used to distinguish cardiovirus strains of enteroviruses from those that are not virulent. In addition, the presence of a virus in a patient with DCM does not necessarily lead to the development of the disease.
Determining viral DNA or RNA using polymerase chain reaction (PCR) is a more reliable method for diagnosing viral myocarditis.
Unfortunately, obtaining myocardial tissue is an invasive method, so not all children can be offered.
Autoimmune reaction
In approximately 30-40% of adult patients with DCM, organ-specific and disease-specific autoantibodies are determined.The absence of these antibodies in other patients may be due to the stage of disease progression.
It has been suggested that a disease, such as viral myocarditis, initiates an autoimmune process with an immune response caused by a superantigen. This process leads to massive activation of T-lymphocytes and myocardial damage.
Genetic predisposition
Genetic causes of the development of congestive cardiomyopathy in children account for 25-50% of cases. The role of genetic factors is confirmed by research on familial DKM. In patients with familial DKM, an increased frequency of the human leukocyte antigen (HLA) -DR4 is observed. It has been reported that the frequency of HLA-DQA1 0501 alleles is significantly higher in patients with idiopathic DKM.

A healthy heart and a patient for stagnant cardiomyopathy
Symptoms of childhood congestive failure
Dilated cardiomyopathy (DKM) refers to dilatation and systolic dysfunction of the ventricles (mainly on the left side) with or without congestive heart failure. This is the most common form of heart muscle disease in children.
The onset of congestive cardiomyopathy usually proceeds imperceptibly, but it can be acute, like in 25% of sick children.Approximately 50% of children had antecedent viral diseases.
The initial symptoms are usually :
- Cough.
- Bad appetite.
- Irritability or unmotivated capriciousness.
- Confused breathing.
- Pallor of the skin.
- Increased sweating.
- Fast fatiguability.
- No weight gain.
- Reducing the amount of urine.
Wheezing, inadequate urination and lack of weight gain is a particularly important clinical sign indicating congestive heart failure in infants.
Other symptoms found in approximately 20% of sick children are as follows:
- Chest pain.
- Palpitations.
- Ortopnea.
- Hemoptysis.
- Foamy sputum.
- Abdominal pain.
- Syncope.
- Neurological disorders.
Video: Cardiomyopathy signs and symptoms | Circulatory System and Disease | NCLEX-RN | Khan Academy
Diagnosis of congestive cardiomyopathy in children
Echocardiography and dopplerography are the main ways to diagnose DKM. These are the most informative non-invasive tests for diagnosing the type of cardiomyopathy and the degree of dysfunction of the heart muscle.
A chest x-ray can reveal cardiomegaly and pulmonary edema. Cardiomegaly, which is accidentally detected on a chest radiograph, or an arrhythmia, as determined on an electrocardiogram (ECG), may be the basis for the initial treatment of the heart.

An ECG often helps in determining the degree of increase in the left ventricle, also showing any abnormal heart rhythm.
Laboratory tests - most often used is a complete blood count, erythrocyte sedimentation rate and C-reactive protein level, which may indicate acute inflammation in children with DKM, especially with the concomitant development of active myocarditis.
MUGA-scanning (multiple-pass arteriography) helps to measure contractile volumes of the left and right ventricles and cardiac output. Refers to non-invasive diagnostic methods.
Invasive procedures
Invasive procedures, such as cardiac catheterization, should be performed by experienced pediatric cardiologists and only when absolutely necessary. Children with DCM are at particular risk of complications during cardiac catheterization and angiography studies.
The main indications for the implementation of invasive procedures:
- Preparing for a heart transplant.
- The need for myocardial biopsy.
Myocardial biopsy is usually done in preparation for heart monitoring after transplantation and follow-up monitoring.
Treatment of congestive cardiomyopathy in children
There are several treatment strategies: pharmacological and surgical in the form of implanting a device.
Pharmacological therapy
Initially, the use of medications for congestive cardiomyopathy in children is mainly focused on the symptoms of heart failure. In particular, are used:
- Diuretics.
- Angiotensin-converting enzyme (ACE) inhibitors.
- Beta blockers.
Diuretics can improve symptoms, whereas ACE inhibitors, most often, prolong the life of the patient. Treatment with beta-blockers in children with chronic heart failure due to DCM has been shown to improve symptoms and left ventricular ejection fraction.

Device implantation
Automatic implantable cardioverter-defibrillators (ICD) reduce the risk of sudden death. Their effectiveness has been clearly demonstrated in adults with chronic congestive heart failure. However, their use in children is somewhat limited.
If necessary, cardiac resynchronization therapy can be performed with AV-synchronous biventricular stimulation.According to the results of some studies, it has been successful in some children with DCM and blockade of the left bundle of His.
Currently, heart transplantation is the optimal treatment for DCM-induced resistant chronic heart failure in children.
Conclusion
Congestive cardiomyopathy in children develops most often after myocarditis, autoimmune disease, or against the background of genetic predisposition. In some cases, the condition does not significantly worsen, but in other cases, clinical manifestations of the type of loss of appetite, chest pain, cough, etc. are observed.
For diagnosis, echocardiography, electrocardiography, and radiography are most often used. In extreme cases, cardiac catheterization and myocardial biopsy are performed. The treatment is based primarily on taking medication, and if they are ineffective, the device is implanted.
Video: Pharmacology - ANTIARRHYTHMIC DRUGS (MADE EASY)
Similar articles
Among all cardiovascular diseases, cardiomyopathy is distinguished in a special way. Pathology can develop not only in adults, but also in children. In the latter case, there are specific features of the course of the disease, its diagnosis and treatment, which should be known to loving parents.

Metabolic disorders can lead to metabolic cardiomyopathy. This disease can significantly disrupt the habitual lifestyle of a person, and in severe cases - lead to death. Therefore, at the slightest sign of a malfunction of the heart, an urgent need to consult a doctor.

Frequent cases when in absolute, at first glance, health a person suddenly dies. In this case, we can talk about physically trained young people, like Jesse Marunda (American athlete) and Miklos Feher (Hungarian football player). In both victims, as in many similar situations, hypertrophic cardiomyopathy was determined, which caused their death.
